|
In his experiment, Moseley shot a stream of electrons at atoms of different elements. It would take until 1913, when British physicist Henry Moseley (1887–1915) began experiments exploring the newly discovered nucleus of the atom. It turns out that some of the imperfections in the Mendeleev periodic table stemmed from using atomic mass as his way of ordering the elements. While Mendeleev’s table was mostly successful, a few elements did not seem to make sense in the positions he assigned. In groups of eight, he observed that as mass increased, so did the number of oxygen atoms that would react with a given element. One of the main patterns Mendeleev observed was the repeating pattern of reactivity with oxygen. 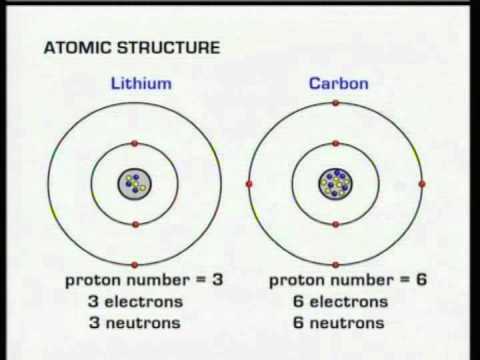
Perhaps most remarkable of all, Mendeleev did all this without knowing the structure of the atom.īecause the proton, the neutron, and the electron had not yet been discovered, Mendeleev initially sorted the elements by atomic mass. It is also important to note that not only did Mendeleev predict the existence of these missing elements, but he also properly predicted their physical and chemical properties. Elements, including germanium and boron, fit neatly into Mendeleev’s table. Later discoveries proved that his predictions were correct. The most remarkable aspect of Mendeleev’s periodic table, and the reason why his name has endured, is that Mendeleev left spaces open for elements that were not yet discovered. Out of the early crop of periodic tables, Russian chemistry professor Dmitri Mendeleev’s table emerged as the defining document of the elements. By the mid-19th century, chemists across the world rushed to organize the elements into a chart that would help them make sense of what they were observing in their experiments. Investigating this further, Döbereiner found that other elements followed this pattern, and so he began to group elements into “triads.” Other chemists built on Döbereiner’s work to observe that elements with similar atomic masses also exhibited similar properties. In the early 19th century, a scientist named Johann Döbereiner (1780–1849) noticed that strontium’s atomic mass fell exactly between the atomic masses of calcium and barium. © The Regents of the University of California, Lawrence Berkeley National Laboratory, 2010. Dmitri Mendeleev created the Periodic Table That We Still Use Today Americans won the rights to name element 104 after Ernest Rutherford (rutherfordium), and the Russians won the rights to name element 105 (dubnium) after Dubna, the town in Russia where the element was discovered.įigure 4-2. Only after the end of the Cold War, in 1997, did IUPAC settle the dispute between the Americans and Soviets. The International Union of Pure and Applied Chemistry, or IUPAC, the global organization charged with establishing standard practices for chemists, is responsible for handing out naming rights to labs in honor of discovering the element. In the 1960s, research groups in the Soviet Union and the United States simultaneously claimed the discovery of elements 104 and 105. The naming of elements turned out to be one of the last battlegrounds of the Cold War. The periodic table contains a wealth of information, once we know how to interpret it. The discoverers of these elements named them in honor of home countries or to commemorate other influential scientists. Some elements have countries in their names (francium, polonium, and germanium), while others bear the names of notable scientists (einsteinium, nobelium, and curium). In addition to chemical properties, the periodic table reveals the cultural history of each element. We also will examine how these patterns allow us to predict the formation of compounds. In this unit, we will discuss some of those patterns, including electron configuration, size, and reactivity. In addition to the atomic number, there are many patterns within the periodic table. 
In fact, if the number of protons was to change, the atom’s identity would change. We can organize the periodic table this way because all atoms of a specific element have the same number of protons. The table is organized by atomic number, which is the number of protons in the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
