|
So therefore I can say molecular formula MAS is equal to end into empirical formula Moss. 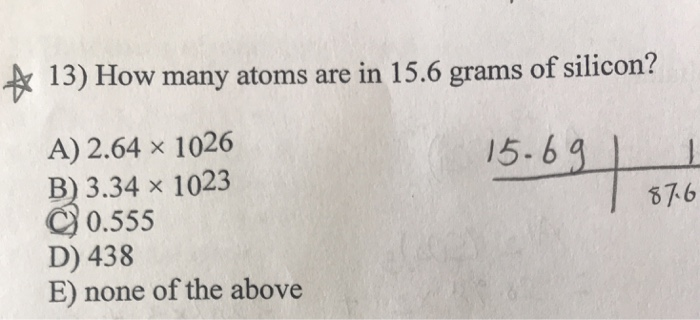
Now the molecular formula MAS that is 64 point 07 graham caramel that is given. So 64 g Permal is the empirical formula Mazz. March, so the empirical formula which is cell for dioxide sa do so empirical formula MAs is equal to 32 for self worth And two into 16. Let us first calculate the empirical formula. So let us first calculate the molecular formula. So with the help of the mamas and the empirical formula we can calculate the molecular formula. Now coming to the question, we have given the mamas and the empirical formula. And we can also write molecular formula mass, that is equal to N into empirical formula moss. So molecular formula is equal to end into empirical formula Where N is the number starting from 1, 2, 3 and up to so on. 
Oh so that means there is a relation between the molecular formula. So glucose molecular formula is C six H 12 or six. So basically the empirical formula shows the ratio of uh the number of items of different elements in one molecule of the compound, while the actual number of different type of item present in one molecule of the compound is the molecular formula for example. So empirical formula is basically the simplest formula and molecular formula is the actual formula of the compound paint. So here the concept is used as the empirical formula and the molecular formula. And we need to calculate the molecular formula of this compound. That is given in the question and the empirical formula that is S. Everyone in this question a compound has Mollema 64.07 g per mole.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
